





Assumptions of Kinetic Theory of Gases
Gas laws
Assuming permanent gases to be ideal, through experiments, it was established that gases irrespective of their nature obey the following laws.
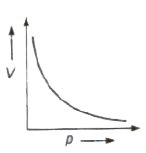
Boyle’s Law
At constant temperature the volume (V) of given mass of a gas is inversely proportional to its pressure (p), i.e.,
V ∝ 1/p ⇒ pV = constant
For a given geas, p1V1 = p2V2
Charles’ Law
At constant pressure the volume (V) of a given mass of gas is directly proportional to its absolute temperature (T), i.e.,
V ∝ T ⇒ V / T = constant
For a given gas, V1/T1 = V2/T2
At constant pressure the volume (V) of a given mass of a gas increases or decreases by 1/273.15 of its volume at 0°C for each 1°C rise or fall in temperature.
Volume of the gas at t°Ce
Vt = V0 (1 + t/273.15)
where V0 is the volume of gas at 0°C.
Gay Lussacs’ or Regnault’s Law
At constant volume the pressure p of a given mass of gas is directly proportional to its absolute temperature T, i.e. ,
p ∝ T ⇒ V/T = constant
For a given gas,
p1/T1 = p2/T2
At constant volume (V) the pressure p of a given mass of a gas increases or decreases by 1/273.15 of its pressure at 0°C for each l°C rise or fall in temperature.
Volume of the gas at t°C, pt = p0 (1 + t/273.15)
where P0 is the pressure of gas at 0°C.
Avogadro’s Law
Avogadro stated that equal volume of all the gases under similar conditions of temperature and pressure contain equal number molecules. This statement is called Avogadro’s hypothesis. According Avogadro’s law
(i) Avogadro’s number The number of molecules present in 1g mole of a gas is defined as Avogadro’s number.
NA = 6.023 X 1023 per gram mole
(ii) At STP or NTP (T = 273 K and p = 1 atm 22.4 L of each gas has 6.023 x 1023 molecules.
(iii) One mole of any gas at STP occupies 22.4 L of volume.
Standard or Perfect Gas Equation
Gases which obey all gas laws in all conditions of pressure and temperature are called perfect gases.
Equation of perfect gas pV=nRT
where p = pressure, V = volume, T = absolute temperature, R = universal gas constant and n = number of moles of a gas.
Universal gas constant R = 8.31 J mol-1K-1.
Real Gases
Real gases deviate slightly from ideal gas laws because
Real or Van der Waal’s Gas Equation
(p + a/V2) (V – b) = RT
where a and b are called van der Waals’ constants.
Pressure due to an ideal gas is given by
p = (1/3).(mn/V). c2 = 1/3 ρ c2
For one mole of an ideal gas
P = (1/3).(M/V).c2
where, m = mass of one molecule, n = number of molecules, V = volume of gas, c = (c12 + c22 + … + cn2) / n allde root mean square (rrns) velocity of the gas molecules and M = molecular weight of the gas. If p is the pressure of the gas and E is the kinetic energy per unit volume is E, then
p = (2/3).E
Kinetic Energy of a Gas
(i) Average kinetic energy of translation per molecule of a gas is given by
E = (3/2) kt
where k = Boltzmann’s constant.
(ii) Average kinetic energy of translation per mole of a gas is given by
E = (3/2) Rt
where R = universal gas constant.
(iii) For a given gas kinetic energy
E ∝ T ⇒ E1/E2 = T1/T2
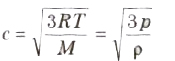
(iv) Root mean square (rms) velocity of the gas molecules is given by
(v) For a given gas c ∝ √T
(vi) For different gases c ∝1/√M
(vii) Boltzmann’s constant k = R/N
where R is ideal gas constant and N = Avogadro number.
Value of Boltzmann’s constant is 1.38 x 10-28 J/K.
(viii) The average speed of molecules of a gas is given by
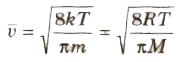
(ix) The most probable speed of molecules of a gas is given by
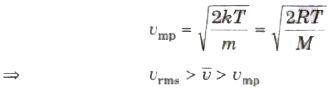

Degree of Freedom
The degree of freedom for a dynamic system is the number of directions in which it can move freely or the number of coordinates required to describe completely the position and configuration of the system.
It is denoted by for N.
Degree of freedom of a system is given by
f or N = 3A – R
where A = number of particles in the system and R = number of independent relations.
Degree of Freedom
Specific heat of a gas
(a) At constant volume, CV = f/2 R
(b) At constant pressure, cp = (f/2 + 1)R
(c) Ratio of specific heats of a gas at constant pressure and at constant volume is given by
γ = 1 + 2/f
Mean Free Path
The average distance travelled by a molecule between two successive collisions is called mean free path (γ).
Mean free path is given by
γ = kT / √2 π σ2p
where σ = diameter of the molecule, p = pressure of the gas,
T = temperature and k = Botlzmann’s constant.
Mean free path
λ ∝ T and λ ∝ 1/p
Brownian Motion
The continuous random motion of the particles of microscopic size suspended in air or any liquid, is called Brownian of microscopic motion.
Brownian suspended motion in both is observed with many liquids and gases.
Brownian motion is due to the unequal bombardment of the suspended Particles by the molecules of the surrounding medium.
.png)